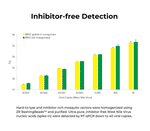
Successfully Added to Cart
Customers also bought...
-
 DNA/RNA Shield (50 ml)Cat#: R1100-50DNA/RNA Shield reagent is a DNA and RNA stabilization solution for nucleic acids in any biological sample. This DNA and RNA stabilization solution preserves the...
DNA/RNA Shield (50 ml)Cat#: R1100-50DNA/RNA Shield reagent is a DNA and RNA stabilization solution for nucleic acids in any biological sample. This DNA and RNA stabilization solution preserves the... -
 DNA/RNA Shield SafeCollect Swab Collection Kit (1 ml fill) (1 collection kit)Cat#: R1160The DNA/RNA Shield SafeCollect Swab Collection Kit is a user-friendly collection kit for stabilizing the nucleic acid content of samples collected with a swab. DNA/RNA...
DNA/RNA Shield SafeCollect Swab Collection Kit (1 ml fill) (1 collection kit)Cat#: R1160The DNA/RNA Shield SafeCollect Swab Collection Kit is a user-friendly collection kit for stabilizing the nucleic acid content of samples collected with a swab. DNA/RNA...
Format
Quick-RNA Viral Kit
Highlights
- Viral RNA: Compatible with plasma, serum, urine, cell culture media, blood, saliva, cellular suspensions, biopsies, swabs, feces, samples in DNA/RNA Shield, etc.
- Streamlined Workflow: Sample inactivation and easy one-step lysis enables fast processing.
- High Sensitivity: Optimized for low viral copy detection for Next-Gen Sequencing and RT-qPCR.
Original Manufacturer
100% satisfaction guaranteed, read Our Promise
Innovated in California, Made in the USA
Format
Quick-RNA Viral Kit
Highlights
- Viral RNA: Compatible with plasma, serum, urine, cell culture media, blood, saliva, cellular suspensions, biopsies, swabs, feces, samples in DNA/RNA Shield, etc.
- Streamlined Workflow: Sample inactivation and easy one-step lysis enables fast processing.
- High Sensitivity: Optimized for low viral copy detection for Next-Gen Sequencing and RT-qPCR.
Original Manufacturer
100% satisfaction guaranteed, read Our Promise
Innovated in California, Made in the USA
| Cat # | Name | Size | Price | Quantity |
|---|
| Cat # | Name | Size | Price | |
|---|---|---|---|---|
| R1200-25 | DNA/RNA Shield (2X Concentrate) | 25 ml | $74.00 | |
| R1200-125 | DNA/RNA Shield (2X Concentrate) | 125 ml | $263.00 | |
| W1001-10 | DNase/RNase-Free Water | 10 ml | $23.00 | |
| W1001-4 | DNase/RNase-Free Water | 4 ml | $14.00 | |
| R1034-1-100 | Viral RNA Buffer | 100 ml | $181.00 | |
| R1034-1-50 | Viral RNA Buffer | 50 ml | $94.00 | |
| R1034-2-48 | Viral Wash Buffer (concentrate) | 48 ml | $127.00 | |
| R1034-2-6 | Viral Wash Buffer (concentrate) | 6 ml | $22.00 | |
| C1001-50 | Collection Tubes | 50 Pack | $18.00 | |
| C1004-50 | Zymo-Spin IC Columns | 50 Pack | $64.00 | |
Description
Performance
Technical Specifications
| Equipment | Microcentrifuge |
|---|---|
| Purity | RNA is ready for Next-Gen sequencing, RT-qPCR, microarray, hybridization, etc. |
| Registration Status | CE-IVD certified (R1034-E, R1035-E) |
| Sample Source | Plasma, serum, saliva, urine, blood, cell culture media, cellular suspensions, biopsies, and swab and fecal samples stored in DNA/RNA Shield |
| Yield | 10 µg RNA (binding capacity), ≥6 µl (elution volume) |
Resources
Documents
FAQ
The purpose of beta-mercaptoethanol is to help with deproteination. It is not necessary if you are working with simple samples such as swabs. It is recommended if you are working with protein rich samples such as plasma, serum, blood, saliva, sputum, etc.
Yes, these samples are compatible.
Yes, this kit will co-purify some DNA.
Most downstream application methods for viral detection do not require DNase treatment during purification. If necessary, DNase treatment can be perform post-purification and additional components can be purchased separately (i.e., DNase I, DNA Digestion Buffer, RNA Prep Buffer and RNA Wash Buffer).
Yes, the change from clear to yellow is a result of oxidation and will not affect the performance of the buffer.
The Viral RNA Buffer is guaranteed for one year from the date of purchase even with the addition of beta-mercaptoethanol. Just be sure to close the bottle cap tightly to prevent evaporation.
In most cases, it could be because the samples contain high amounts of protein or cellular debris. To help prevent this in future preps, it’s best to add beta-mercaptoethanol to the Viral RNA buffer, and/or implement a Proteinase K digestion step.
For optimal results and detection of viral target, collect sample in DNA/RNA Shield and perform Proteinase K treatment. In addition, add beta-mercaptoethanol to the Viral RNA Buffer prior to purification, as well as perform all steps at room temperature and centrifugation speeds at 10,000-16,000 x g to ensure no buffer retention.
The “-E" markings at the end of the catalog numbers of “-Dx” products indicate that they are CE-IVD verified for in vitro diagnostic use.
This can happen on occasion due to transport or storage at lower temperatures. The reagent functionality is not affected; however, the precipitate can be resolved by heating the reagent to >37 °C.
Reviews
"The Quick-RNA Viral RNA kit performed much better than the RNeasy 96 kit for extracting RNA from dilute samples of virus supernatant. The kit is designed for small-volume elution to automatically concentrate the sample, and it’s designed for viral supernatants rather than cell lysates, so we didn’t need to deal with optimizing added carrier RNA. So in all, it was simpler, easier, and gave us a 10-fold improvement in our limit of detection."
S.B.
Boehringer Ingelheim, Canada
Citations
Need help? Contact Us